Hoku defends his thesis entitled Regulation of Non-muscle Myosin II. Congratulations, Dr. West-Foyle!... Read More
To See the World in a Dividing Cell
If you are interested in supporting our research or our Johns Hopkins Initiative for Careers in Science and Medicine and the Summer Academic Research Experience program for socioeconomically under-resourced youth, please contact Doug (dnr@jhmi.edu), Kevin McGuire, Sr. Associate Director of Development (kmcgui10@jhmi.edu), or Sarah Farrell, Director of Development (sfarrell@jhmi.edu), Institute for Basic Biomedical Sciences.
Lab announcements:
Mar. 13, 2024: Check out this cool new video describing our research – How Contractility Kits Can Be Used to Fight Cancer. This beautiful 3D animation was created by Art as Applied to Medicine master’s student, Nick Kilner-Pontone.
Dec. 16, 2023: Paul Trevorrow at Cytoskeleton kindly interviewed Doug about his journey in science. You can read the interview here: An interview with Douglas N. Robinson—Johns Hopkins School of Medicine, Baltimore, Maryland, USA
Dec. 1, 2023: We welcome our new doctoral student, Carter Swaby, to the lab. Welcome aboard, Carter! Thanks for letting us join in your journey!
July 27, 2023: Congratulations, Eleana, for receiving the HHMI Gilliam Fellowship.
June 5, 2023: Check out the new writeup on our Johns Hopkins Initiative for Careers in Science and Medicine.
April 25, 2023: We welcome our new doctoral student, Barbara Talavera Figueroa, to the lab. Welcome, Barbara! We are excited to have you along on our journey!
Feb. 13, 2023: We welcome our new SARE Director and Lab Manager, Pamela Hamm. Welcome, Pam, and we are very excited to get to work with you on our many big vision efforts!
Dec. 23, 2022: Doctoral student Ly Nguyen just completed her doctoral degree. She is beginning a new position at Intellia Therapeutics. Big things are on the horizon, Ly! Have fun!
Oct. 14, 2022: Doctoral student Shantel Angstadt just completed her doctoral degree and is now starting as an iCURE postdoctoral fellow at NIH. Shantel originally joined us as an undergraduate REU student in 2013, and now she is Dr. Angstadt! Thanks for letting us be a part of your journey, and warm wishes for you in this next exciting phase, Shantel!
Sept. 5, 2022: Doctoral student Yinan Liu just defended her thesis. Yinan is off to Flagship Pioneering where she will be an Associate. We wish her a lot of fun and success in her new exciting adventure in venture capital and biotech startup world!
June 30, 2022: Amoebas, Worms and Flies, Oh My! Check out this write up describing the power of model organisms for generating insights into human disease. We appreciate that our work using Dictyostelium to generate insights into intractable diseases such as pancreatic cancer is highlighted.
May 27, 2022: Our lab family member Jessica Leng is heading off today to start her new adventure as a medical student at Case Western Reserve. This will be an exciting next phase, Jessica!
Apr 22, 2022: We welcome our new doctoral student, Brooke Waechtler, to the lab. Brooke, welcome to our team!
Jul 23, 2021: Check out my interview by Dr. Kimberly Skarupski for the Faculty Factory podcast series. Here we discussed setting up and operating a research lab and the importance of mentorship in the process: Habits and Hacks with Douglas N. Robinson, PhD
Jun 15, 2021: Our work just got featured in Scientific American: Treating a Deadly Lung Disease with a Little Help from Amoebas. Here is the Scientific American issue.
May 7, 2021: We welcome our new doctoral students, Eleana Parajón and Mark Allan Jacob. Eleana and Mark, I am excited about all the discoveries we will make together!
Apr 13, 2021: Check out our latest press release on how “Amoeba biology reveals a potential treatment target for lung disease.”
Feb 26, 2021: Congrats to Corrine and Jenny for their awesome work described in our new paper on adenine nucleotide translocase as a protector from cigarette smoke and its important role in airway hydration and ciliary function. Check out Corrine’s First Person interview about this work here.
Sep 29, 2020: Our patent for Treating and Preventing Diseases by Modulating Cell Mechanics (US Patent: US10787410B2) has been issued.
Sep 24, 2020: Doug Robinson gets recognized as an ASCB Fellow. Fellows are selected for their lifetime achievements in advancing cell biology.
Jul 24, 2020: Thinzar Htwe, SARE Scholar 2019 and rising Stanford freshman, published her excellent Op-Ed piece on the importance of socioeconomic and racial/ethnic diversity in STEM and healthcare. Her Op-Ed was published in the Stanford Daily: Preparing Future Generations: BIPOC and FLI representation in science, research, and medicine.
Jan 20, 2020: Doctoral Diversity Program Scholar and now 2nd-yr medical student Michelle Colbert and I got to share the story of the Johns Hopkins Initiative for Careers in Science and Medicine on “Midday with Tom Hall on WYPR (88.1) in Baltimore. Michelle was awesome in this interview! Here is the link: MLK Jr. Day of Service, Pt 2: Hopkins’ Careers in Science & Medicine Initiative
Dec 2, 2019: Thanks Ashanti Edwards and Ahna Skop for inviting us to prepare a piece for the ASCB newsletter in which we describe the Johns Hopkins Initiative for Careers in Science and Medicine.
Nov 29, 2019: Check out the interviews with three Summer Academic Research Experience (SARE) alums: Q&A — The Next Generation of Scientific Researchers.
Nov 22, 2019: November is COPD Awareness Month. Chronic Obstructive Pulmonary Disease is currently the 3rd leading cause of disease-associated death in the U.S., and yet no therapeutic strategy directly addresses the root cause of the disease. To address complex diseases like COPD requires an interdisciplinary team, and thankfully we have had the opportunity to build such a team that includes two pulmonary physician scientists Ramana Sidhaye and Corrine Kliment and a pharmacology doctoral student Jenny Nguyen. Thank you, Biophysical Society, for sharing our work on your blog.
Oct 17, 2019: Check out the new Technology Feature by Vivien Marx in Nature Methods. Vivien highlights some of the breakthroughs and impact in the mechanobiology field. Thanks, Vivien, for writing this!
Sept 18, 2019: Check out the press release on our new Cancer Research paper: Johns Hopkins Researchers ID Compound That Could Play A Novel Role In Halting Pancreatic Cancer Progression
May 24, 2019: Our lab’s mission – the galaxy depends on us! 🙂
Apr 9, 2019: Congratulations to Priyanka for winning the David Yue Award as part of the School of Medicine’s Young Investigator’s Day recognition! Read Priyanka’s YIDP interview here.
Jan 16, 2019: Check out Priyanka’s First Person Interview and the journal cover art published with her article in Journal of Cell Science! Great job, Priyanka!
May 15, 2018: Doug was awarded the Provost’s Prize for Faculty Excellence in Diversity.
Jun 2, 2017: You can listen to Doug’s ASBMB Ruth Kirschstein Diversity of Science Award talk here.
Jun 2, 2017: See Doug’s ASBMB interview for his recognition by ASBMB’s Ruth Kirschstein Diversity of Science Award.
Feb 24, 2017: Check out the Baltimore Sun article on our new DARPA-funded project. We are aiming to engineer Dictyostelium cells to be able to perform specific tasks.
Feb 19, 2017: Check out our video describing our outreach program, the Johns Hopkins Initiative for Careers in Science and Medicine.
Feb 2, 2017: Doug is selected as a Science Super Hero by Discovery Communications. The goal of the program is to call attention to the impact of science on community. We thank Discovery Communications for their recognition of our efforts.
Dec 12, 2016: Check out our Op-Ed piece in support of the federal Health Careers Opportunity Program: Don’t cut a federal program that helps disadvantaged students enter health careers. Please remind your senators and representatives how much impact we can have for a pretty small investment!
Mar 4, 2016: Corrine Kliment was just awarded the Baurenschmidt Award from the Eudowood Board. Congrats, Corrine! You can read more about Corrine’s work in this ASCB write up.
Dec 14, 2015: Please check out our ASCB Celldance Video. We attempted to present how you go from basic science discovery to making an impact on a disease, in this case on pancreatic cancer. The Celldance Videos are designed to communicate our science stories through live cell imaging for a general audience. The press release may be found here.
Aug 22, 2015: Check out this video on our 4-HAP work on ScIQ, TYT’s New Science Channel: Enjoy!
Jun 10, 2015: Check out Mariya Khan’s animatic depicting the design constraints of a dividing cell. Please make sure your sound is turned on.
See our earlier press releases:
Wanted: Self-Driving Cells to Pursue Deadly Bacteria: Johns Hopkins Team Aims to Make Micro-Soldiers That Seek Out and Subdue Pathogens
Drawing on their expertise in control systems and cell biology, Johns Hopkins University researchers are setting out to design and test troops of self-directed microscopic warriors that can locate and neutralize dangerous strains of bacteria. Please see the full story here.
Cellular ‘Cruise Control’ Systems Let Cells Sense and Adapt to Changing Demands
Cells are the ultimate smart material. They can sense the demands being placed on them during critical life processes and then respond by strengthening, remodeling or self-repairing, for instance. To do this, cells use “mechanosensory” systems similar to the cruise control that lets a car’s engine adjust its power output when going up or down hills. Researchers are uncovering new details on cells’ molecular cruise control systems. By learning more about the inner workings of these systems, scientists hope ultimately to devise ways to tinker with them for therapeutic purposes. Please check out the rest of the story on National Institute of General Medical Science’s BioBeat.
Proteins Pull Together As Cells Divide: Group dynamics, not star proteins, drive mechanics of crucial cell process
Like a surgeon separating conjoined twins, cells have to be careful to get everything just right when they divide in two. Otherwise, the resulting daughter cells could be hobbled, particularly if they end up with too many or two few chromosomes. Successful cell division hangs on the formation of a dip called a cleavage furrow, a process that has remained mysterious. Now, researchers at Johns Hopkins have found that no single molecular architect directs the cleavage furrow’s formation; rather, it is a robust structure made of a suite of team players. This work appeared online in Current Biology on February 19, 2015. Please click here for the rest of the story.
Under Pressure: Mechanical stress is a key driver of cell-cell fusion, study finds
Just as human relationships are a two-way street, fusion between cells requires two active partners: one to send protrusions into its neighbor, and one to hold its ground and help complete the process. Researchers have now found that one way the receiving cell plays its role is by having a key structural protein come running in response to pressure on the cell membrane, rather than waiting for chemical signals to tell it that it’s needed. The study, which helps open the curtain on a process relevant to muscle formation and regeneration, fertilization, and immune response, appears in the March 9 (2015) issue of the journal Developmental Cell. Please click here for the rest of the story.
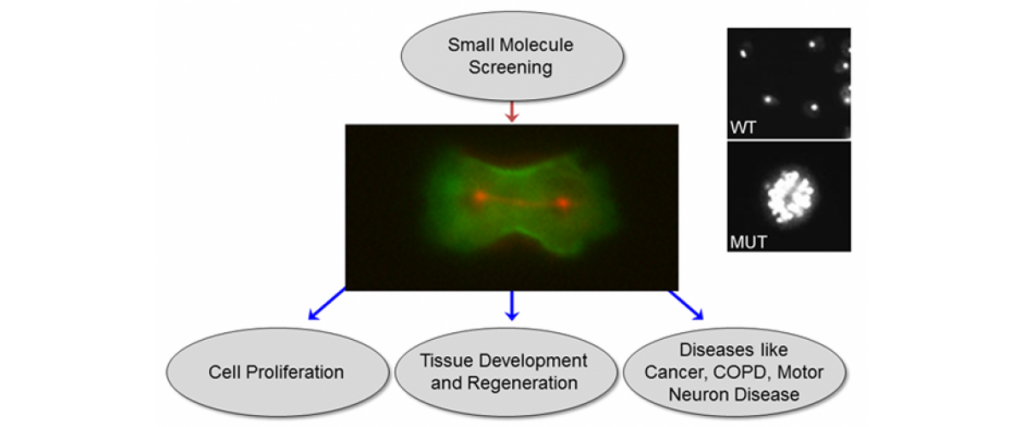
Stiffening Up Cancer Cells
A new screen uncovers compounds that alter cell mechanics. Could these compounds someday treat cancer from an unexpected angle? Find out by reading the story in Biotechniques.
New Cancer-Fighting Strategy Would Harden Cells to Prevent Metastasis
Existing cancer therapies are geared toward massacring tumor cells, but Johns Hopkins researchers propose a different strategy: subtly hardening cancer cells to prevent them from invading new areas of the body. They devised a way of screening compounds for the desired effect and have identified a compound that shows promise in fighting pancreatic cancer. Their study appeared January 20, 2015 in the early edition of the Proceedings of the National Academy of Sciences. Please click here for the rest of the story.
My Lab’s Commitment to Diversity, Equity, and Inclusion may be found in this document: Robinson Lab DEI Statement.
My Lab’s Training Outcomes:
A new coalition of 9 top universities and an institute calls for transparency in the outcomes and demographics of every institution’s training programs (here is the Science Magazine writeup). We do not need a mandate to provide that information. Here are my lab’s trainee outcomes and demographics:
To date, 101 members have passed through our lab, including 21 doctoral students, 6 postdoctoral fellows, 1 clinical research fellow, 33 undergraduate and medical students, 10 technicians and SARE directors/lab managers, 2 Chemical and Biomolecular Engineering master’s student, 4 Art as Applied to Medicine masters students, and 27 outreach and high school students. Two students spent summers as undergrads in the lab then returned for their doctoral training. The 101 includes 68 (67%) women and 28 (28%) Underrepresented in Science (UIS) or Medicine (UIM). We have also had 5 visiting scientists spend extended periods of time in the lab.
My doctoral students complete their degrees in an average of 5.6 years (median 5.6 years) and produce an average of 5.5 papers (range 2-12; mean of 2.6 first author). My postdoctoral fellows have completed in an average of 4.1 years (median 2.8 years) and produced an average of 4.4 papers (range 1-13; mean of 2 first author).
Example career outcomes of my doctoral and postdoctoral trainees include tenure track faculty (42% of postdocs, approximately 3-times the national rate), science writing, patent law, FDA reviewing, venture world (e.g., Deerfield Management, Flagship Pioneering), and scientists in biotech (e.g., Abbvie, Genentech, NextCure, Intellia, and SocialCode).
My undergraduates have gone on to medical school (in one case with a $120,000 scholarship to U. Pittsburgh based on his work in my lab), graduate school (e.g., Northwestern, UC Berkeley, UNC Chapel Hill), and MSTP programs (e.g., Cornell-Rockefeller). In two cases, my summer undergraduate students (one through the Summer Internship Program (SIP) and one (a UIS) from the Research Experience for Undergraduates (REU)) returned to my lab for their doctoral studies.
My high school students have all moved on to college, including students from low-income, educationally under-resourced backgrounds (many of whom are pursuing STEM or health-related degrees) that have come through my lab’s Summer Academic Research Experience (SARE) program (Kabacoff et al. CBE Life Sci. Educ. 2013; http://sare.cellbio.jhmi.edu). Please note that SARE itself has now served 127 high school scholars. 95% of those who have reached college age are confirmed to have matriculated into community college or 4-year college programs. SARE scholars have matriculated into colleges all over the country, including at Hopkins, Stanford, Yale, Princeton, University of Maryland College Park, Howard, LSU, and many, many more. SARE scholars have at least a 59% college completion rate by four years post-high school graduation. By six years post-high school graduation, at least 69% of SARE scholars have graduated from college. This 69% compares quite favorably to the 14% national rate for students from low-income, educationally under-resourced backgrounds, which is assessed by six years post-college matriculation. 79% of SARE scholars graduate with degrees in STEMM (Science, Technology, Engineering, Mathematics, and Medical health professions). From SARE, we created the Johns Hopkins Initiative for Careers in Science and Medicine (CSM). The CSM includes stage-appropriate programs for 5th grade, high school, undergraduate, and post-baccalaureate scholars. To date, more than 635 scholars have participated in the CSM, and our scholars are pursuing a wide range of STEMM careers, including MD, MD/PhD, and PhD degrees, all over the country.